
The proton is made up of two up quarks and one down quark. A neutron is made of two down quarks and one up quark. These are the up quark, the down quark, the strange quark, the charm quark, the bottom quark, and the top quark. Quarks are subatomic particles, but they are also elementary particles because we do not know if they are made up of even smaller particles. Protons and neutrons are made up of quarks. Atoms are the fundamental components of matter. 
In atoms, there is a small nucleus in the center, which is where the protons and neutrons are, and electrons orbit the nucleus. Protons, electrons, and neutrons are the three subatomic particles that typically make up an atom. Elementary particles are quarks, leptons and bosons. Both neutrons and protons are assigned as having masses of 1. The Standard Model has a defined number of key particles: elementary and composite (def). For simplicity, we will use the amu unit for the three subatomics. Masses for the three subatomic particles can be expressed in amu ( atomic mass units) or grams. The electron has a negative charge (a - charge), and it is the smallest of these three particles. Atoms are made up of protons and neutrons located within the nucleus, with electrons in orbitals surrounding the nucleus. The proton has a positive charge (a + charge). These are the particles which make atoms. The proton, the neutron, and the electron are subatomic particles. 1 Particles currently thought to be elementary include electrons, the fundamental fermions ( quarks, leptons, antiquarks, and antileptons, which generally are matter particles and antimatter particles), as. Subatomic particles are particles that are smaller than atoms. In particle physics, an elementary particle or fundamental particle is a subatomic particle that is not composed of other particles. Nanoparticles are an intermediate size, being a very fine powder but much larger than atoms.Ītoms and molecules are called microscopic particles. Powder and dust are some examples of macroscopic particles. Macroscopic particles are particles that are larger than atoms or molecules. There are many different types of particles, with different particle sizes and properties. Constitutional values as stated in the South Africa constitution. Issue relating to citizens rights and responsibilities. Time-management skills and reading and writing for different purposes. In particle physics, an elementary particle is a particle which cannot be split up into smaller pieces. Skilled for Life Sexual behaviour and sexual health Mr Nhavoto. The atom shown here is a helium atom.Ī particle is a tiny bit of matter that makes up everything in the universe. We have included a succinct definition for each of the words, so students will be assimilating new information while they have fun. 2 archaic : a clause or article of a composition or document. b : a relatively small or the smallest discrete portion or amount of something. 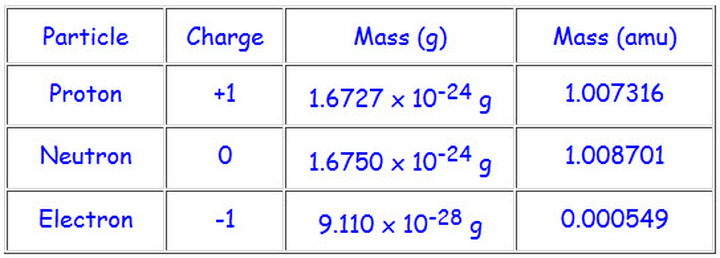 
an extremely small piece of matter that is smaller than an atom or found inside an atom, such as. Browse subatomic particles puzle resources on Teachers Pay Teachers, a marketplace trusted by millions of teachers for original educational resources. What is a particle simple definition 1a : a minute quantity or fragment. Red circles represent protons and blue circles represent neutrons. subatomic particle définition, signification, ce quest subatomic particle: 1. When trains carrying chlorine gas derail.This is an atom, and it is very small. Neutral chlorine atoms instantlyĬombine to form Cl 2 molecules, which are so reactive that entire communities are evacuated Into flame when it comes in contact with water. Sodium metal, for example, which consists of neutral sodium atoms, bursts Has an enormous impact on the chemical and physical properties of the atom. The gain or loss of electrons by an atom to form negative or positive ions By adding one moreĮlectron we get a negatively charged Cl - ion with a net charge of -1. A neutral chlorineĪtom, for example, contains 17 protons and 17 electrons. By removing an electron from this atom we get a positivelyĬharged Na + ion that has a net charge of +1.Ītoms that gain extra electrons become negatively charged. According to modern atomic theory, an atom has a nucleus, which is its center, or core. A neutral sodium atom, for example, contains 11 protonsĪnd 11 electrons. A subatomic particle is a unit of matter or energy that's the fundamental makeup of all matter. Neutral atoms can be turned into positively charged ions by removing one When an ion is formed, the number of protons By definition,Īn ion is an electrically charged particle produced by either removing electronsįrom a neutral atom to give a positive ion or adding electrons to a neutralĪtom to give a negative ion. Atoms are neutral they contain the same number of protons as electrons. Atoms are the tiny particles of an element that are responsible for chemical reactions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
